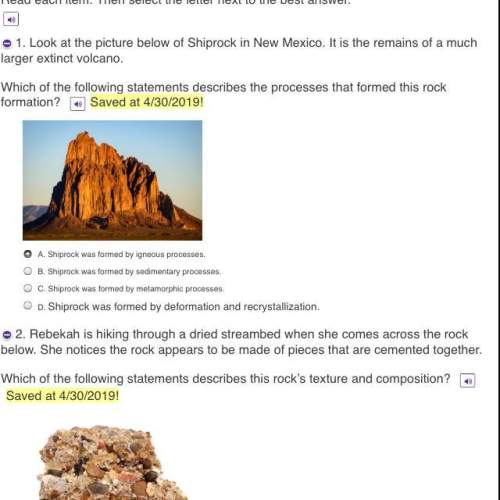
1.changes to a substance that do not involve a permanent reordering of atoms are know as
physical changes.
endothermic changes.
irreversible changes.
chemical changes.
2.changes that reorder atoms to form new molecules, such as the reaction between sodium and chlorine ions to form sodium chloride, are known as
irreversible changes.
boiling point changes.
chemical changes.
physical changes.
3.what does the release or absorption of energy indicate?
chemical change
neither physical nor chemical changes
physical change
physical and chemical changes
4.which of these is an example of a chemical change?
melting cheese
ironing a shirt
fireworks exploding
grinding powder
5.which of the following changes can alter the form of a substance but does not necessarily change it to an other substance?
physical changes
pressure changes
temperature changes
all of the above
6.chemical compounds undergo both physical and chemical changes. which of the following is an example of a chemical change
combining oxygen gas and hydrogen gas to form liquid water
salt (nacl) being dissolved into water
heating a solid material to a liquid form
cooling a gas into its liquid form
7.in physical science lab, kim and dawn added small pieces of magnesium to hydrochloric acid. they observed that bubbles formed, the test tube got hot, and the magnesium disappeared. what is a sign that a chemical reaction has taken place in this experiment?
magnesium appeared
temperature decreased
gas is produced
odor
8.if two poisonous elements are combined chemically, which of the following will be true of the resulting compound?
the compound may or may not be poisonous, depending on the chemical properties of the new substance.
the compound will not be poisonous, based on the chemical properties of the new substance.
the compound will be more poisonous than the elements, based on the chemical properties of the two elements.
the compound will be as poisonous as the elements, based on the chemical properties of the two elements.
9.students are studying how two substances react. the students mix substance 1 with substance 2 in a solution. later the students boil the solution, releasing substance 1 as a gas and leaving substance 2 as a solid.
this is a chemical change, because the two substances were mixed together.
what type of change is described in the paragraph above? how do you know?
this is a chemical change, because the two substances were separated chemically by boiling.
this is a physical change, because the two substances were separated physically by boiling.
this is a chemical change, because a gas was released.
10.max drops one effervescent tablet into a beaker of water and places an identical tablet on the lab table. he observes bubbles of gas form on the surface of the first tablet. the bubbles rise and break on the surface of the water. after several minutes, no more bubbles form and the tablet is not visible in the water. the tablet on the lab table is unchanged. what conclusion should max draw from his observations?
the tablet that was placed in the water broke down when exposed to light and formed a gas through a chemical change.
the tablet that was placed in the water changed directly into a gas through a physical change.
the tablet that was placed in the water interacted with the water to form a gas through a chemical change.
the tablet that was placed in the water caused dissolved gases in the water to be released through a physical change.
11.which of these is true of a chemical reaction?
a. energy is always released.
b. energy is either released or absorbed.
c. energy is always absorbed.
d. energy is not transferred.
12.you are given two jars, each of which contains an unknown substance. you are asked to determine if the substances are different from one another or if each jar contains the same substance. what should you do to complete this task?
13.which of these is a chemical property of a substance?
a. flammability
b. boiling point
c. density
d. color
14.atoms in the same of the periodic share the same chemical properties.
column
side
group (row)
state of matter
15.which of the following are physical properties? check all that apply.
conductivity
flammability
melting point
reactivity with water
solubility
combustibility
malleability
density
16.which of the following represent chemical properties? check all that apply.
odor
oxidation
flammability
conductivity
solubility
combustibility
density
boiling point
17.what characteristic of an atom determines its properties?
a. arrangement of protons in the nucleus
b. the atomic mass
c. the arrangement of electrons around the nucleus
d. the atomic number of neutrons
i need

Answers: 3
Another question on Biology

Biology, 22.06.2019 03:00
Discuss the functions of epithelial connective nerviud and muscular tissues
Answers: 3


Biology, 22.06.2019 13:00
The smallest parts of these that retain their original properties are called
Answers: 1

Biology, 22.06.2019 15:30
Me.henley said, “in the rat race we call life, only the strong will survive.” he may have been referring to
Answers: 1
You know the right answer?
1.changes to a substance that do not involve a permanent reordering of atoms are know as
Questions


Arts, 09.10.2019 08:50

Mathematics, 09.10.2019 08:50

Mathematics, 09.10.2019 08:50



Mathematics, 09.10.2019 08:50


Chemistry, 09.10.2019 08:50

English, 09.10.2019 08:50

Mathematics, 09.10.2019 08:50

English, 09.10.2019 08:50






Biology, 09.10.2019 09:00

English, 09.10.2019 09:00

Health, 09.10.2019 09:00