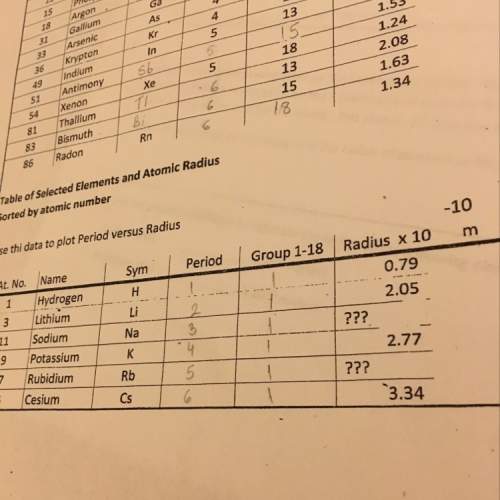
Chemistry, 21.07.2019 02:00 lorenzo1beaton
Will mark as brainliest. atomic radius generally increases as we move down a group and from right to left across a period up a group and from left to right across a period up a group and from right to left across a period explain your answer.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 20:30
If 10.g of agno3 is available, what volume of 0.25 m agno3 can be prepared
Answers: 1

Chemistry, 22.06.2019 05:30
What type of reaction is shown below? check all that apply. 2h2o2 → 2h2o + o2 synthesis decomposition combustion
Answers: 1

Chemistry, 22.06.2019 11:00
Which statement correctly identifies the scientific question and describes why the question is scientific? question 1 refers to the supernatural.question 2 reflects a moral or social value.question 3 refers to something that can be measured.question 4 reflects a question that can’t be observed.
Answers: 1

Chemistry, 22.06.2019 11:50
The chemical bond connecting one nucleotide with the next one along the nucleic acid chain is called a
Answers: 3
You know the right answer?
Will mark as brainliest. atomic radius generally increases as we move down a group and from right t...
Questions




World Languages, 02.12.2020 20:00

Mathematics, 02.12.2020 20:00

Biology, 02.12.2020 20:00

Mathematics, 02.12.2020 20:00


English, 02.12.2020 20:00

Spanish, 02.12.2020 20:00


Mathematics, 02.12.2020 20:00

Mathematics, 02.12.2020 20:00


Business, 02.12.2020 20:00



Mathematics, 02.12.2020 20:00

Health, 02.12.2020 20:00