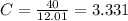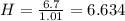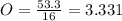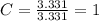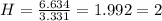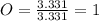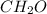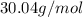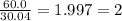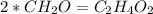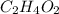Answers: 1
Another question on Chemistry



Chemistry, 22.06.2019 06:00
If a polyatomic ionic compound has gained two hydrogen ions, then how does its name begin?
Answers: 3

Chemistry, 22.06.2019 10:30
Which of these is not an example of chemical weathering? a. iron-rich mineral rusting b. feldspar turning into clay c. limestone reacting with acid d. granite breaking up into sand
Answers: 1
You know the right answer?
An unknown compound is found to contain 40.0 percent carbon, 6.7 percent hydrogen and 53.3 percent o...
Questions


Mathematics, 03.08.2019 01:00





Mathematics, 03.08.2019 01:00

History, 03.08.2019 01:00





Mathematics, 03.08.2019 01:00



Mathematics, 03.08.2019 01:00

Social Studies, 03.08.2019 01:00



English, 03.08.2019 01:00