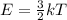Chemistry, 17.07.2019 04:30 thegreentnt5025
You are experimenting on the effect of temperature on the rate of reaction between hydrochloric acid (hcl) and potassium iodide (ki). when the reaction is completed at 273k there are approximately 250,000 collisions per mole of reactant. you run the experiment again at 350k. which of the following would you expect to be the number of collisions recorded at 350k a. 2000 b. 700,000 c. 0 d. 200,000

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 15:10
Which statement describes the phase change that occurs when dry ice is placed in an open container at room temperature?
Answers: 1


Chemistry, 23.06.2019 09:00
The vapor pressure of water at 25.0°c is 23.8 torr. determine the mass of glucose (molar mass = 180 g/mol) needed to add to 500.0 g of water to change the vapor pressure to 22.8 torr.
Answers: 1

You know the right answer?
You are experimenting on the effect of temperature on the rate of reaction between hydrochloric acid...
Questions




Mathematics, 15.12.2021 01:50

Mathematics, 15.12.2021 01:50

Mathematics, 15.12.2021 01:50

Business, 15.12.2021 01:50

Mathematics, 15.12.2021 01:50

Mathematics, 15.12.2021 01:50


History, 15.12.2021 01:50

Mathematics, 15.12.2021 01:50



English, 15.12.2021 01:50