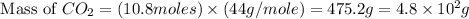Consider the reaction between acetylene, c2h2, and oxygen in a welding torch: 2c2h2(g) + 5o2(g) → 4co2(g) + 2h2o(g) if 5.4 moles of acetylene react with sufficient oxygen, how many grams of co2 will be formed? a. 2.4 × 102 g b. 9.5 × 102 g c. 4.8 × 102 g d. 1.5 × 102 g e. 0.49 g

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
Which of the following statements is true about planck’s law
Answers: 1

Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 1

Chemistry, 22.06.2019 12:10
Achemistry student needs to standardize a fresh solution of sodium hydroxide. he carefully weighs out of oxalic acid , a diprotic acid that can be purchased inexpensively in high purity, and dissolves it in of distilled water. the student then titrates the oxalic acid solution with his sodium hydroxide solution. when the titration reaches the equivalence point, the student finds he has used of sodium hydroxide solution.calculate the molarity of the student's sodium hydroxide solution. be sure your answer has the correct number of significant digits.
Answers: 1

Chemistry, 22.06.2019 22:00
The volume of an unknown substance in a sealed glass jar is 50 milliliters. the volume of the jar is 200 milliliters. which state of matter could the substance be?
Answers: 2
You know the right answer?
Consider the reaction between acetylene, c2h2, and oxygen in a welding torch: 2c2h2(g) + 5o2(g) → 4...
Questions

History, 08.06.2020 04:57


History, 08.06.2020 04:57

Mathematics, 08.06.2020 04:57

Mathematics, 08.06.2020 04:57




History, 08.06.2020 04:57











Social Studies, 08.06.2020 04:57

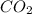 formed will be,
formed will be, 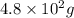
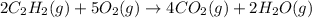
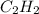 react to give 4 mole of
react to give 4 mole of 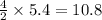 moles of
moles of