Answer : The correct answer is solutions of 0.100 M of Magnesium hydroxide and Beryllium hydroxide.
The Hydroxide ions(OH⁻) are produced when ionic compounds (basic compound ) dissociates in water or compound reacts with water as follows :
MOH (s) + H2O (l) <=> M+ (aq) + OH⁻(aq)
M (aq) + H₂O(l) <=> MH (aq) + OH⁻(aq)
Where MOH and M are compounds.
Concentration of OH⁻ can be found in compound by following steps :
Step 1 : To find acid or base .
First of all we need to find whether the given compound is acid or base by acid - base concept . (image attached )
If a compound is acid , it will not produce OH⁻ ions or negligible amount of OH⁻ ions. So it can be ruled out .
If the compound is base , step 2 should be followed .
Step 2 : To find strength of base .
We need to find whether the given base is strong or weak .
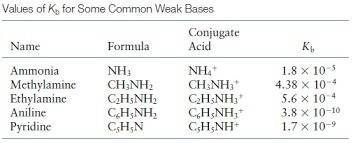
Generally hydroxides of Group 1 and 2 are strong base . Here are some listed strong base ( image attached ) .Base other than these criteria will be weak base .
Step 3 :To find concentration of OH⁻ ions :
If the base is strong , means it will dissociate completely to produce OH⁻ ions . Hence concentration of OH⁻ can be calculated by stoichiometric relation from balanced reaction.
If base is weak , it will not dissociate completely to give all the OH⁻ ions , so we will make ICE table of a balanced reaction for those base and use kb values to calculate concentration of base .
A) Rubidium hydroxide (RbOH ) (0.100 M ) :
According to Arrhenius concept RbOH is a base and from list it is a strong base . Hence it will dissociate completely to give OH⁻ ions .
RbOH + H₂O <=> Rb⁺ + OH⁻
Since the Ratio of RbOH : OH⁻ = 1 : 1 .
![[OH^-] = 0.100 M RbOH * \frac{1 OH^-}{1 RbOH } = 0.100 M OH^-](/tpl/images/0257/1263/4e4f0.png)
[OH⁻] = 0.100 M
B) Magnesium hydroxide , Mg(OH)₂ ( 0.100 M) :
According to Arrhenius concept and since Mg is from group 2 , so Mg(OH)₂ can be said as Strong base . So Mg(OH)₂ will dissociate completely as :
Mg(OH)₂ + H₂O <=> Mg²⁺ + 2 OH⁻
The ratio of OH⁻ : Mg(OH)₂ = 2 : 1
![[OH^- ] = 0.100 M Mg(OH)2 * \frac{2 OH^-}{1 Mg(OH)2}](/tpl/images/0257/1263/05d7d.png)
[OH⁻ ] = 0.200 M
C) Ammonia (NH3 ) (0.100 M ) :
According to Lewis concept it is base but it neither belongs to list nor group 1 and 2 , Hence it is weak base and will dissociate incompletely as :
NH₃ + H₂O <=> NH₄OH <=> NH₄⁺ + OH⁻
Since it is weak base so ICE table will be formed as:
NH₃ + H₂O <=> NH₄⁺ + OH⁻
Initial(M) 0.100 , - 0 0
Change(M) -x - +x + x
Equilibrium(M) 0.100-x - x x
Now Kb expression will be written as :
![kb = \frac{[NH4^+] * [OH^-] }{[NH3]}](/tpl/images/0257/1263/46c69.png)
Kb for NH3 is 1.8 * 10 ⁻⁵ ( from list in image )
Plugging equilibrium values of [NH₄⁺] = [OH⁻] = x and [NH₃] = (0.100-x ) in this expression :
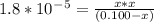
Since kb is small (10⁻⁵ ) , x from (0.100 -x ) can be neglected
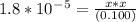
Multiplying both side by 0.100
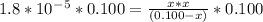
x² = 1.8 * 10⁻⁶
x = 1.34 * 10⁻³
Hence concentration of OH⁻ produced by NH₃ = 1.34 * 10⁻³ M
D) Beryllium hydroxide ,Be(OH)₂ (0.100 M ) :
According to Arrhenius concept it is a base and since it belongs to Group 2 , its is strong base . Hence it will dissociate completely .
Be(OH)₂ + H₂O <=> Be²⁺ + 2 OH⁻
Ratio between OH⁻ : Be(OH)₂ = 2 : 1
![[OH^- ] = 0.100 M Be(OH)2 *\frac{2 OH^-}{1 Be(OH)2}](/tpl/images/0257/1263/25ebd.png)
Hence [OH⁻] by Be(OH)₂ = 0.200 M
E) Hydrochloric acid , HCl (0.100 M ) :
According to all the concepts HCl is acid . Since it is acid it produce negligible amount of OH⁻ ion . So this option can be neglected .
If all are summarized :
[OH⁻] by RbOH = 0.100 M
[OH⁻] by Mg(OH)₂ = 0.200 M
[OH⁻] by NH₃ = 1.34 * 10⁻³ M
[OH⁻] by Be(OH)₂ = 0.200 M
[OH⁻] by HCl = negligible
Hence Mg(OH)₂ and Be(OH)2 produced highest amount of [OH⁻] .





























![[OH^-] = 0.100 M RbOH * \frac{1 OH^-}{1 RbOH } = 0.100 M OH^-](/tpl/images/0257/1263/4e4f0.png)
![[OH^- ] = 0.100 M Mg(OH)2 * \frac{2 OH^-}{1 Mg(OH)2}](/tpl/images/0257/1263/05d7d.png)
![kb = \frac{[NH4^+] * [OH^-] }{[NH3]}](/tpl/images/0257/1263/46c69.png)
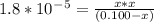
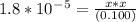
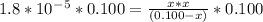
![[OH^- ] = 0.100 M Be(OH)2 *\frac{2 OH^-}{1 Be(OH)2}](/tpl/images/0257/1263/25ebd.png)




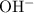 :
: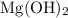 :
: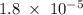
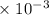 M
M 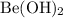 ;
; 

