

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Match the mixture with the substance// i really need on this guys (it’s a pic btw)
Answers: 1

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3

Chemistry, 23.06.2019 02:10
Detrimental the length of the object shown 1. 97.8mm 2. 97.80 mm 3. 97mm 4. 98mm
Answers: 2

Chemistry, 23.06.2019 03:50
How many moles of potassium are needed to react completely with 12.8 moles of magnessium bromide?
Answers: 2
You know the right answer?
A92.8 gram sample of cuso4•5h2o.(copper sulfate pentahydrate) is heated until water is released. how...
Questions

Physics, 02.10.2020 14:01


Mathematics, 02.10.2020 14:01


Mathematics, 02.10.2020 14:01




History, 02.10.2020 14:01

Health, 02.10.2020 14:01

Chemistry, 02.10.2020 14:01


Mathematics, 02.10.2020 14:01

Social Studies, 02.10.2020 14:01

Mathematics, 02.10.2020 14:01

Chemistry, 02.10.2020 14:01



Mathematics, 02.10.2020 14:01

Mathematics, 02.10.2020 14:01

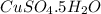
![[63.55+32+(4\times 16)+5(16+(2\times 1))]=249.55g/mol](/tpl/images/0088/5520/3a76e.png)
![[16+(2\times 1)]=18g/mol](/tpl/images/0088/5520/7a48c.png)
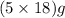 of water molecule is present.
of water molecule is present.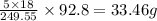 of water molecule.
of water molecule.

