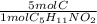Chemistry, 14.07.2019 07:00 hannahgrace325
Valine is an amino acid with the formula c5h11no2. determine the number of moles of carbon in 99.82 g of valine.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 01:30
In a spacecraft, the following reaction occurs: co2(g) + 2lioh(s) -> lico3(s) + h2o(i) (i attached picture of equation) how many liters of carbon dioxide will 4 moles of lithium hydroxide (lioh) absorb? (one mole of any gads occupies 22.4 l under certain conditions of temperature and pressure. assume those conditions for this equation.) 45l 6.0l 3.0l 34l
Answers: 1

Chemistry, 22.06.2019 16:00
About 3% of the water on earth is freshest. only about 40% of that freshwater is available for human use. why is so much freshwater unavailable for human use?
Answers: 2

Chemistry, 22.06.2019 18:00
Chlorophyll a had the molecular formula c55h72mgn4o5 how many atoms are in this molecule
Answers: 2

Chemistry, 22.06.2019 21:20
40dm3 of gas at 760 torr are heated from 5°c to 50°c what is the new volume
Answers: 3
You know the right answer?
Valine is an amino acid with the formula c5h11no2. determine the number of moles of carbon in 99.82...
Questions



Social Studies, 12.12.2019 20:31


History, 12.12.2019 20:31



Mathematics, 12.12.2019 20:31



History, 12.12.2019 20:31



Chemistry, 12.12.2019 20:31

Mathematics, 12.12.2019 20:31


Mathematics, 12.12.2019 20:31

Biology, 12.12.2019 20:31

English, 12.12.2019 20:31

 ×
× ×
×