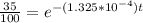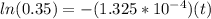Chemistry, 13.07.2019 18:30 joshuajoseph249
The half-life of a radioactive isotope is the amount of time it takes for a quantity of that isotope to decay to one half of its original value. (a) express the half-life of a radioactive isotope in terms of its decay rate. (b) the half-life of radiocarbon or carbon 14 (c-14) is 5230 years. determine its decay rate parameter . (c) carbon dating is a method of determining the age of an object using the properties of radiocarbon. it was pioneered by willard libby and collaborators in 1949 to date archaeological, geological, and other samples. its main idea is that by measuring the amount of radiocarbon still found in the organic mater and comparing it to the amount normally found in living matter, we can approximate the amount of time since death occurred.7 using the decay-rate parameter found in part (b), nd the time since death if 35% of radiocarbon is still in the sample.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 22:00
If a plot weight (in g) vs. volume (in ml) for a metal gave the equation y= 13.41x and r^2=0.9981 what is the density of the metal?
Answers: 2

Chemistry, 22.06.2019 09:00
Acrystal that absorvd water from air is (blank)a. aqueousb. homogenousc. hygroscopicd. efflorescent
Answers: 1

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 12:30
A50.0 ml sample of gas at 20.0 atm of pressure is compressed to 40.0 atm of pressure at constant temperature. what is the new volume? 0.0100 ml 0.325 ml 25.0 ml 100. ml
Answers: 1
You know the right answer?
The half-life of a radioactive isotope is the amount of time it takes for a quantity of that isotope...
Questions

Social Studies, 28.07.2021 16:10

Mathematics, 28.07.2021 16:10

Spanish, 28.07.2021 16:10

Mathematics, 28.07.2021 16:10





Geography, 28.07.2021 16:10

History, 28.07.2021 16:10



Mathematics, 28.07.2021 16:10

Computers and Technology, 28.07.2021 16:10




Physics, 28.07.2021 16:10

Mathematics, 28.07.2021 16:10

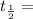
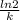
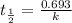
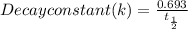
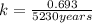

![[A] = [A_{0}]e^{-kt}](/tpl/images/0085/7348/51547.png)
![\frac{[A]}{[A_{0}]} = e^{-kt}](/tpl/images/0085/7348/f4d8d.png)