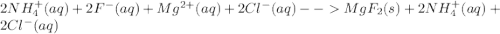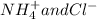Write the net ionic equation for this reaction occurring in water: ammonium fluoride and magnesium chloride are mixed to form magnesium fluoride and ammonium chloride. 1. no reaction occurs. 2. 2 nh+ 4 + 2 cl− → 2 nh4cl 3. 2 f− + mg2+ → mgf2 4. 2 nh4f + 2 cl− → 2 f− + 2 nh4cl 5. 2 f− + mgcl2 → mgf2 + 2 cl−

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
What determines the average kinetic energy of the particles in a gas? a. the number of collisions b. the number of particles c. the size of the particles d. the temperature
Answers: 1


Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2

Chemistry, 22.06.2019 19:00
What is the compound name for the formula [ru(en)2cl2]2+ and [co(en)cl2br]-
Answers: 1
You know the right answer?
Write the net ionic equation for this reaction occurring in water: ammonium fluoride and magnesium...
Questions

History, 25.08.2020 20:01



English, 25.08.2020 20:01



Social Studies, 25.08.2020 20:01



English, 25.08.2020 20:01

History, 25.08.2020 20:01




Biology, 25.08.2020 20:01



Social Studies, 25.08.2020 20:01


Mathematics, 25.08.2020 20:01

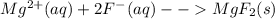

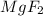 will not split into ions as it is insoluble in water.
will not split into ions as it is insoluble in water.