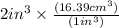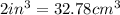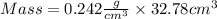The mass unit associated with density is usually grams. if the volume (in ml or cm3) is multiplied by the density (g/ml or g/cm3) the volume units will cancel out, leaving only the mass units. keep in mind that the volume and density must use the same volume unit for the cancellation. if a large marshmallow has a volume of 2.00 in3 and density of 0.242 g/cm3, how much would it weigh in grams? 1 in3=16.39 cm3. express your answer in grams to three significant figures.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:00
In which of these cases are the two wave points considered to be in phase with each other?
Answers: 1


Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 02:30
What role does weathering have in shaping earth’s surface? a) it allows sediments to fall out of a medium. b) it sediments settle on a new surface. c) it breaks down older material into sediments. d) it transports sediments to a different location. will give brainliest, answer quickly.
Answers: 2
You know the right answer?
The mass unit associated with density is usually grams. if the volume (in ml or cm3) is multiplied b...
Questions

English, 28.01.2021 19:30



Mathematics, 28.01.2021 19:30

Mathematics, 28.01.2021 19:30

Advanced Placement (AP), 28.01.2021 19:30

Social Studies, 28.01.2021 19:30

Spanish, 28.01.2021 19:30




Mathematics, 28.01.2021 19:30

Mathematics, 28.01.2021 19:30


Mathematics, 28.01.2021 19:30


Arts, 28.01.2021 19:30

History, 28.01.2021 19:30


Mathematics, 28.01.2021 19:30

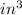
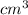
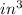 and unit of volume in density is
and unit of volume in density is 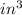 to
to