Chemistry, 12.07.2019 09:30 pedavis6609
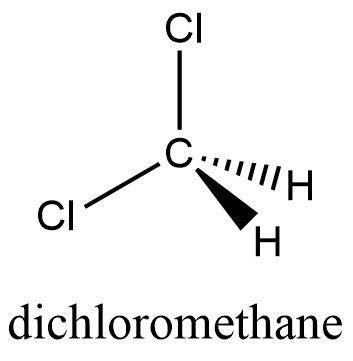
Amolecule contains five atoms and has a molecular weight of 85 g per mole. the atoms are of elements with atomic numbers 1, 6, and 17. which molecular structure could represent this molecule?

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:00
What is the approximate vapor pressure when the gas condenses at 70 degrees celsius
Answers: 2

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1

Chemistry, 22.06.2019 23:50
Be sure to answer all parts. the following equilibrium constants were determined at 1123 k: c(s) + co2(g) ⇌ 2co(g) k'p = 1.30 × 1014 co(g) + cl2(g) ⇌ cocl2(g) k''p = 6.00 × 10−3 calculate the equilibrium constant at 1123 k for the reaction: c(s) + co2(g) + 2cl2(g) ⇌ 2cocl2(g) 4.68 × 10 9 (enter your answer in scientific notation.) write the equilibrium constant expression, kp:
Answers: 3

Chemistry, 23.06.2019 00:00
What conclusion can you draw from this experiment about the components of the black ink?
Answers: 3
You know the right answer?
Amolecule contains five atoms and has a molecular weight of 85 g per mole. the atoms are of elements...
Questions



Social Studies, 31.03.2020 19:46

Mathematics, 31.03.2020 19:46

Biology, 31.03.2020 19:46

English, 31.03.2020 19:46

English, 31.03.2020 19:48


Mathematics, 31.03.2020 19:49



English, 31.03.2020 19:50



Mathematics, 31.03.2020 19:50

Mathematics, 31.03.2020 19:50

Chemistry, 31.03.2020 19:50

English, 31.03.2020 19:50