
Chemistry, 12.07.2019 09:30 beccadrums
Gadolinium oxide, a colorless powder which absorbs carbon dioxide from the air, contains 86.76 mass % gd. determine its empirical formula.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:00
Asolution contains 225 g of sodium chloride, nacl, dissolved in enough water to make a 0.25 l of solution. what is the molarity of the solution?
Answers: 2

Chemistry, 22.06.2019 05:50
Why doesn't heat added to water make the tempature rise above 100c
Answers: 2


Chemistry, 23.06.2019 06:40
The combustion of methane, ch4, releases 890.4kj/mol. that is, when one mole of methane is burned,890.4 kj are given off to the surroundings. this meansthat the products have 890.4 kj less than the reactants.thus, ah for the reaction = - 890.4 kj. a negative symbolforah indicates an exothermic reaction.ch (g) + 20 (g)> co2 (g) + 2 h0 (1); ah = - 890.4 kga) how much energy is given off when 2.00 mol of ch,are burned? b) how much energy is released when 22.4g of ch. areburned?
Answers: 1
You know the right answer?
Gadolinium oxide, a colorless powder which absorbs carbon dioxide from the air, contains 86.76 mass...
Questions


Mathematics, 16.01.2020 18:31







Biology, 16.01.2020 18:31



Mathematics, 16.01.2020 18:31


Mathematics, 16.01.2020 18:31


Mathematics, 16.01.2020 18:31

Mathematics, 16.01.2020 18:31

Geography, 16.01.2020 18:31


English, 16.01.2020 18:31

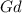 = 86.76 % (given)
= 86.76 % (given)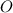 = 100 - 86.76 = 13.24 %
= 100 - 86.76 = 13.24 %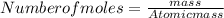
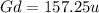
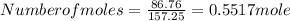
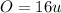
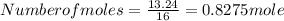
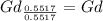
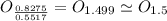
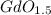 , will multiply by 2 to get the whole integer.
, will multiply by 2 to get the whole integer.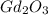 .
.

