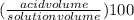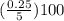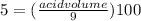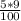Chemistry, 11.07.2019 22:30 twentyonepilots12018
Achemist has 5 l of a solution that contains 0.25 l of acetic acid. the chemist needs to prepare 9 l of a solution with the same percent concentration of acetic acid. how much acidic acid must the 9 l of the solution contain

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1


Chemistry, 21.06.2019 22:40
How many electrons does silver have to give up in order to achieve a sido noble gas electron configuration
Answers: 1

Chemistry, 22.06.2019 03:50
Consider the reaction: n2(g) + o2(g) ? 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3
You know the right answer?
Achemist has 5 l of a solution that contains 0.25 l of acetic acid. the chemist needs to prepare 9 l...
Questions







Computers and Technology, 24.12.2019 02:31