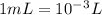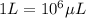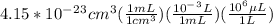Chemistry, 11.07.2019 15:30 daniel2humnle
The volume of a single strontium atom is 4.15×10-23 cm3. what is the volume of a strontium atom in microliters

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:10
Nitric oxide (no) can be formed from nitrogen, hydrogen and oxygen in two steps. in the first step, nitrogen and hydrogen react to form ammonia: n2(g) + 2 h_2(g) rightarrow 2 nh_3 (g) delta h = -92. kj in the second step, ammonia and oxygen react to form nitric oxide and water: 4 nh_3(g) + 5 o_2(g) rightarrow 4no(g) + 6 h_2o(g) delta h = -905. kj calculate the net change in enthalpy for the formation of one mole of nitric oxide from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 1

Chemistry, 22.06.2019 23:10
Match the formula for the following compound: magnesium sulfate heptahydratemgs·7h2omg2so4·7h2omg(so4)2·7h2omgso4·7h2o
Answers: 1

Chemistry, 23.06.2019 03:00
Achemical equilibrium between gaseous reactants and products is shown. n2(g) + 3h2(g) ⇌ 2nh3(g) how will the reaction be affected if the pressure on the system is increased? it will shift toward the reactant side as there is lower pressure on the reactant side. it will shift toward the product side as there is higher pressure on the product side. it will shift toward the reactant side as there are a greater number of moles of gas on the reactant side. it will shift toward the product side as there are a fewer number of moles of gas on the product side.
Answers: 2

Chemistry, 23.06.2019 03:30
Ahelium balloon contains 16.9 l of helium at stp. how many atoms of helium are in the balloon
Answers: 1
You know the right answer?
The volume of a single strontium atom is 4.15×10-23 cm3. what is the volume of a strontium atom in m...
Questions

Mathematics, 22.01.2021 20:40



Mathematics, 22.01.2021 20:40

English, 22.01.2021 20:40


Biology, 22.01.2021 20:40


Spanish, 22.01.2021 20:40

Geography, 22.01.2021 20:40



Mathematics, 22.01.2021 20:40

History, 22.01.2021 20:40

Mathematics, 22.01.2021 20:40

Mathematics, 22.01.2021 20:40

Computers and Technology, 22.01.2021 20:40

Computers and Technology, 22.01.2021 20:40

Chemistry, 22.01.2021 20:40

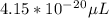
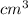 to microliters.
to microliters. 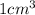 = 1 mL
= 1 mL