
Solid lead(ii) iodide was prepared by reacting 65.0 ml of a solution containing 0.218 m lead(ii) ions with 80.0 ml of a solution containing 0.265 m iodide ions. if the actual yield of the reaction was 3.26 g, which choice is closest to the %yield of the reaction?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 21:30
If i make a solution by adding 83grams of sodium hydroxide to 750ml i’d water what is the molarity of sodium hydroxide
Answers: 1

Chemistry, 23.06.2019 02:00
The bone of a dinosaur and the imprint of a leaf are examples of which kind of fossils? a) index b) body c) amber d) trace
Answers: 1

Chemistry, 23.06.2019 02:50
Dumbledore decides to gives a surprise demonstration. he starts with a hydrate of na2co3 which has a mass of 4.31 g before heating. after he heats it he finds the mass of the anhydrous compound is found to be 3.22 g. he asks everyone in class to determine the integer x in the hydrate: na2co3·xh2o; you should do this also. round your answer to the nearest integ
Answers: 2

Chemistry, 23.06.2019 09:00
Individuals within populations exhibit some diversity. as a result of possessing slightly different traits, some individuals are better able to survive and reproduce than others. if these individuals changes in the characteristics of the population may occur over time. the cumulative change in these characteristics is known as
Answers: 3
You know the right answer?
Solid lead(ii) iodide was prepared by reacting 65.0 ml of a solution containing 0.218 m lead(ii) ion...
Questions







Business, 18.11.2020 17:10



Mathematics, 18.11.2020 17:10



Advanced Placement (AP), 18.11.2020 17:10

History, 18.11.2020 17:10






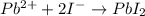
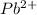 is 0.218 M and 65 mL respectively. Convert it into number of moles as follows:
is 0.218 M and 65 mL respectively. Convert it into number of moles as follows: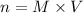
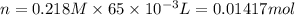
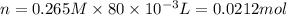
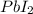 , thus, 0.01417 mol will give 0.01417 mol of
, thus, 0.01417 mol will give 0.01417 mol of 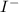 will give 1 mole of
will give 1 mole of 
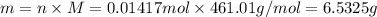
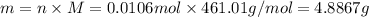
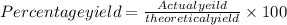
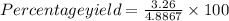 =66.7%
=66.7%

