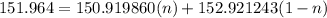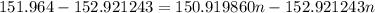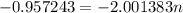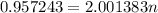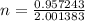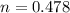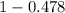Chemistry, 10.07.2019 03:30 dependentclause5828
The atomic masses of 151eu and 153eu are 150.919860 and 152.921243 amu, respectively. the average atomic mass of europium is 151.964 amu. calculate the natural abundances of these two isotopes.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 17:10
Calculate the estimated density of each ball. use the formula d = m/v where d is the density, m is the mass, and v is the volume. record your calculations in table a of your student guide. given that the density of water is 1.0 g/cm3, make a prediction about whether each ball will float in water. record your prediction in table a. what is the estimated density of the table tennis ball? record your answer to the nearest hundredth
Answers: 2

Chemistry, 23.06.2019 01:30
In which phase of mitosis do the spindle fibers pull the chromosomes apart to opposite sides of the cell ?
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1

Chemistry, 23.06.2019 10:30
Me soon im confused much mass would a mole of hydrogen molecules contain? recall that hydrogen is diatomic. g/mol
Answers: 1
You know the right answer?
The atomic masses of 151eu and 153eu are 150.919860 and 152.921243 amu, respectively. the average at...
Questions




Mathematics, 07.07.2019 14:20


History, 07.07.2019 14:20

Business, 07.07.2019 14:20

History, 07.07.2019 14:20

History, 07.07.2019 14:20


Chemistry, 07.07.2019 14:20



Chemistry, 07.07.2019 14:20

History, 07.07.2019 14:20


History, 07.07.2019 14:20

Biology, 07.07.2019 14:20


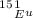 is 0.478 or 47.8% and
is 0.478 or 47.8% and 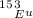 is 0.522 or 52.2% .
is 0.522 or 52.2% .