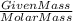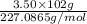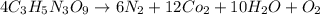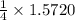Chemistry, 09.07.2019 17:00 mathwiznot45
Be sure to answer all parts. nitroglycerin (c3h5n3o9) is a powerful explosive. its decomposition may be represented by 4c3h5n3o9 → 6n2 12co2 10h2o o2 this reaction generates a large amount of heat and gaseous products. it is the sudden formation of these gases, together with their rapid expansion, that produces the explosion. (a) what is the maximum amount of o2 in grams that can be obtained from 3.50 × 102 g of nitroglycerin

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 12:20
Which is an example of the practical pursuit of alchemy? a. forming perfect substances. b. transforming base metals. c. developing metalworking techniques. d. linking spiritual characteristics with material substances.
Answers: 1

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2
You know the right answer?
Be sure to answer all parts. nitroglycerin (c3h5n3o9) is a powerful explosive. its decomposition may...
Questions

Social Studies, 14.12.2020 21:40

Physics, 14.12.2020 21:40


Mathematics, 14.12.2020 21:40






Mathematics, 14.12.2020 21:40


Chemistry, 14.12.2020 21:40


Mathematics, 14.12.2020 21:40




Mathematics, 14.12.2020 21:40


Health, 14.12.2020 21:40

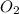 obtained = 12.576 g
obtained = 12.576 g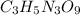 = 3.50 × 102g
= 3.50 × 102g