
Chemistry, 09.07.2019 04:00 alexcuevaz90
For the reaction shown, identify the element oxidized, the element reduced, the oxidizing agent, and the reducing agent. kno3 → kno2 + o2

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 09:30
In apex! a liquid heated beyond a certain temperature becomes
Answers: 1

Chemistry, 22.06.2019 14:30
100 grams of molten lead (600°c) is used to make musket balls. if the lead shot is allowed to cool to room temperature (21°c), what is the change in entropy (in j/k) of the lead? (for the specific heat of molten and solid lead use 1.29 j/g⋅°c; the latent heat of fusion and the melting point of lead are 2.45 × 104 j/kg and 327°c, respectively.)
Answers: 1

Chemistry, 23.06.2019 02:00
As light moves from one material into the next, which of the following affects how much the light waves will refract, or bend? angle at which the ray strikes the medium color of the material density of the material temperature of the light wave
Answers: 2

Chemistry, 23.06.2019 09:20
La reaccion entre monoxido de nitrogeno (no) y oxigeno para formardioxido de nitrogeno (no2) es un paso determinante para la formacion del smog, la reaccion es la siguiente: 2no + o2 = 2no2 cual sera el numero de moles de no2 que se formaran por la reaccion completa de 8 moles de oxigeno con suficiente monoxido?
Answers: 1
You know the right answer?
For the reaction shown, identify the element oxidized, the element reduced, the oxidizing agent, and...
Questions


Mathematics, 12.07.2021 20:40

Mathematics, 12.07.2021 20:40



Mathematics, 12.07.2021 20:40


Mathematics, 12.07.2021 20:40


Mathematics, 12.07.2021 20:40

Arts, 12.07.2021 20:40


Mathematics, 12.07.2021 20:40

Mathematics, 12.07.2021 20:40

Health, 12.07.2021 20:40


Social Studies, 12.07.2021 20:40



Physics, 12.07.2021 20:40

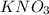 is act as an oxidizing agent as well as reducing agent.
is act as an oxidizing agent as well as reducing agent.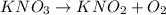
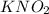 is calculated as:
is calculated as: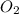 is Zero (o).
is Zero (o).

