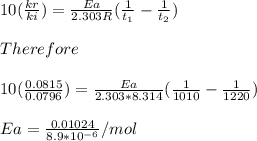Chemistry, 07.07.2019 21:00 madisontrosclair2
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollution generated in automobile engines. the decomposition of nitric oxide (no) to n2 and o2 is second order with a rate constant of 0.0796 m−1⋅s−1 at 737∘c and 0.0815 m−1⋅s−1 at 947∘c. you may want to reference (page) section 14.5 while completing this problem. part a calculate the activation energy for the reaction. express the activation energy in kilojoules per mole to three significant digits.

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:40
*will mark you brainliest + 15 points ** why does the equilibrium of a system shift when the pressure is increased? a. to maximize the stress on the system b. to stop restoring equilibrium to the system c. to increase the total moles of gas in the system d. to decrease the total moles of gas in the system
Answers: 3

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 23.06.2019 00:50
The chemical formula for emerald is be3al2(sio3)6.an emerald can be decided as
Answers: 3

Chemistry, 23.06.2019 01:00
Atoms contain subatomic particles called protons and neutrons. when these protons and neutrons spilt, a lot of energy is released
Answers: 3
You know the right answer?
Understanding the high-temperature behavior of nitrogen oxides is essential for controlling pollutio...
Questions

History, 21.09.2019 09:30


Mathematics, 21.09.2019 09:30

Social Studies, 21.09.2019 09:30

Physics, 21.09.2019 09:30



Mathematics, 21.09.2019 09:30

Biology, 21.09.2019 09:30



Mathematics, 21.09.2019 09:30

Spanish, 21.09.2019 09:30

English, 21.09.2019 09:30

Mathematics, 21.09.2019 09:30

Social Studies, 21.09.2019 09:30

Social Studies, 21.09.2019 09:30



Mathematics, 21.09.2019 09:30