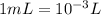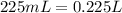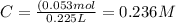Chemistry, 07.07.2019 21:00 jonthedon62
What is the concentration (in m) of a 225ml potassium sulfate solution that contains 4.15g of potassium?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 17:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. initial mass and yield sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 1

Chemistry, 22.06.2019 06:30
Melting and boiling are endothermic processes. this means that these processes absorb energy from their surroundings in order to occur. use this information and the data you collected in the phase change gizmo to describe what happens to the temperature of water when you boil it, then explain why this result occurs.
Answers: 1

Chemistry, 22.06.2019 10:40
Which buffer would be better able to hold a steady ph on the addition of strong acid, buffer 1 or buffer 2? explain. buffer 1: a solution containing 0.10 m nh4cl and 1 m nh3. buffer 2: a solution containing 1 m nh4cl and 0.10 m nh3
Answers: 1

Chemistry, 22.06.2019 16:30
How many grams of mgbr2 are needed to produce 75g or metal?
Answers: 1
You know the right answer?
What is the concentration (in m) of a 225ml potassium sulfate solution that contains 4.15g of potass...
Questions

Biology, 08.04.2020 17:34

History, 08.04.2020 17:34



Mathematics, 08.04.2020 17:34

Biology, 08.04.2020 17:34




Mathematics, 08.04.2020 17:34








English, 08.04.2020 17:34


Social Studies, 08.04.2020 17:34

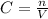 .... (1)
.... (1)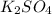 thus, there are 2 moles of potassium in 1 mol of potassium sulfate.
thus, there are 2 moles of potassium in 1 mol of potassium sulfate.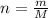
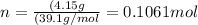
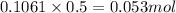 .
.