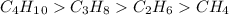Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 06:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 3

Chemistry, 22.06.2019 07:30
The table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 3

Chemistry, 22.06.2019 14:30
In water, a strong acid will break down into its component parts. a. completely b. partly c. never in water, a weak base will break down into its component parts. a. completely b. partly c. never
Answers: 2

Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3
You know the right answer?
Arrange the molecules in order from strongest intermolecular force to weakest. -c2h6 -c4h10 -ch4 -c3...
Questions


Mathematics, 29.08.2019 15:30

Biology, 29.08.2019 15:30

Mathematics, 29.08.2019 15:30


Geography, 29.08.2019 15:30

Biology, 29.08.2019 15:30

History, 29.08.2019 15:30


Biology, 29.08.2019 15:30


Computers and Technology, 29.08.2019 15:30

Geography, 29.08.2019 15:30

Social Studies, 29.08.2019 15:30



Mathematics, 29.08.2019 15:30


History, 29.08.2019 15:30

Mathematics, 29.08.2019 15:30