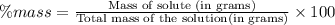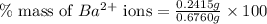Chemistry, 05.07.2019 11:30 pharadorvil04
Asample of 0.6760 g of an unknown compound containing barium ions (ba2+) is dissolved in water and treated with an excess of na2so4. if the mass of the baso4 precipitate formed is 0.4105 g, what is the percent by mass of ba in the original unknown compound?

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Layers of rock containing fossils, like the layers illustrated here, are most likely composed of rocks.
Answers: 2



Chemistry, 23.06.2019 03:50
Show how to convert the temperature 84.7° c to kelvin. include all steps and label the final answer.
Answers: 1
You know the right answer?
Asample of 0.6760 g of an unknown compound containing barium ions (ba2+) is dissolved in water and t...
Questions

Mathematics, 11.10.2019 16:10


Mathematics, 11.10.2019 16:10




English, 11.10.2019 16:10













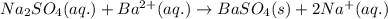
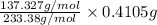 of Barium ions
of Barium ions