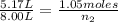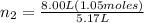Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 02:50
Using a value of ksp = 1.8 x 10-2 for the reaction pbcl2 pb+2(aq) + 2cl -(aq). if the value of ksp was determined to be only 1.2 x 10-2: too much solid has dissolved. additional precipitate is forming. the solution is unsaturated. the ions are now combining to reduce their concentrations.
Answers: 3

Chemistry, 22.06.2019 06:30
Use examples from the article to explain one positive and one negative effect that chemistry has had on society.
Answers: 2

Chemistry, 22.06.2019 20:00
Carbon-14 undergoes radioactive decay in the reaction above. determine the type of radiation emitted in this reaction and describe what is happening to the nucleus during this reaction.
Answers: 2

Chemistry, 23.06.2019 07:30
How do you interpret a chromagram for what mixtures contain?
Answers: 1
You know the right answer?
An inflatable toy starts with 1.05 moles of air and a volume of 5.17 liters. when fully inflated, th...
Questions

English, 14.02.2022 05:40

Mathematics, 14.02.2022 05:40


Mathematics, 14.02.2022 05:40


Mathematics, 14.02.2022 05:40



Mathematics, 14.02.2022 05:40


Mathematics, 14.02.2022 05:40

Biology, 14.02.2022 05:40






Biology, 14.02.2022 05:40

Mathematics, 14.02.2022 05:40

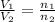
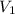 = 5.17 L,
= 5.17 L, 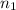 = 1.05 moles
= 1.05 moles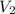 = 8.00 L,
= 8.00 L, 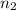 = ?
= ?