Chemistry, 02.07.2019 17:30 bartlettcs9817
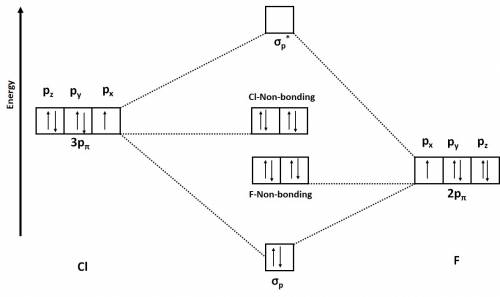
Use the drawing of a molecular orbital energy diagram for clf (assume that the σp orbitals are lower in energy than the π orbitals.) to predict the bond order?

Answers: 1
Another question on Chemistry


Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 06:00
Oxidation-reduction reactions (often called "redox" for short) are reactions that involve the transfer of electrons from one species to another. oxidation states, or oxidation numbers, allow chemists to keep track of these electron transfers. in general, one element will lose electrons (oxidation), with the result that it will increase in oxidation number, and another element will gain electrons (reduction), thereby decreasing in oxidation number. the species that is oxidized is called the reducing agent or reductant. the species that is reduced is called the oxidizing agent or oxidant. to sum up: oxidation = increase in oxidation state = loss of electrons = reducing agent reduction = decrease in oxidation state = gain of electrons = oxidizing agent part a which element is oxidized in this reaction? fe2o3+3co→2fe+3co2 enter the elemental symbol. view available hint(s) is oxidized part b which element is reduced in this reaction? 2hcl+2kmno4+3h2c2o4→6co2+2mno2+2kcl+4h2o enter the elemental symbol. view available hint(s) is reduced
Answers: 1

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3
You know the right answer?
Use the drawing of a molecular orbital energy diagram for clf (assume that the σp orbitals are lower...
Questions




Computers and Technology, 03.02.2020 08:58


English, 03.02.2020 08:58



English, 03.02.2020 08:58

Physics, 03.02.2020 08:58


Business, 03.02.2020 08:58

Mathematics, 03.02.2020 08:58

Mathematics, 03.02.2020 08:58

History, 03.02.2020 08:58

History, 03.02.2020 08:58



Computers and Technology, 03.02.2020 08:58

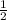 [Electrons in boding orbital - electrons in anti bonding orbital]
[Electrons in boding orbital - electrons in anti bonding orbital]