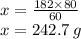Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 04:50
Acompound contains c, h, and o atoms. when 1.130 g of the compound is burned in oxygen, 1.064 g co2 and 0.3631 g h2o are produced. what is the empirical formula of this compound?
Answers: 1

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1


Chemistry, 22.06.2019 11:50
Acompound has a molecular weight of 12.124 atomic mass units and the empirical formula c3h40. what is the molecular formula of the compound?
Answers: 3
You know the right answer?
Sio2 + 4hf → sif4 + 2h2o in this chemical reaction, how many grams of hf are needed for 182 grams of...
Questions

Biology, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00


Mathematics, 18.09.2019 01:00

Mathematics, 18.09.2019 01:00


Biology, 18.09.2019 01:00



Mathematics, 18.09.2019 01:00



Mathematics, 18.09.2019 01:00

Physics, 18.09.2019 01:00