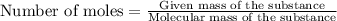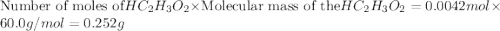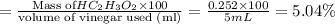The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams of hc2h3o2/ volume of vinegar used (ml) ) x 100. if 5.0 ml of vinegar were used for the titration and 0.0042 moles of hc2h3o2 were required to reach the endpoint, calculate the percent of hc2h3o2 in vinegar. the molar mass of hc2h3o2 is 60.0 g / mol. question 10 options:

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1


Chemistry, 22.06.2019 21:00
Need what is special about water as a compound? how does water regulate climate? what drives water evaporation? why is the water vapor fresh water when it rises from the ocean? why might freshwater in the form of snow take longer to enter the water cycle again than liquid precipitation? what is an aquifer? what role do people play in the water cycle? plz just answer as many as you can ! thx if you !
Answers: 1

Chemistry, 23.06.2019 00:00
This statement about matter and its behavior is best classified as a
Answers: 1
You know the right answer?
The formula to determine the percent of hc2h3o2 (mass/volume) in vinegar is percent (m/v) = (grams o...
Questions


Biology, 29.06.2019 00:00



Mathematics, 29.06.2019 00:00

English, 29.06.2019 00:00

Computers and Technology, 29.06.2019 00:00


Mathematics, 29.06.2019 00:00

Mathematics, 29.06.2019 00:00

Social Studies, 29.06.2019 00:00



Mathematics, 29.06.2019 00:00



Mathematics, 29.06.2019 00:00


Social Studies, 29.06.2019 00:00

Mathematics, 29.06.2019 00:00

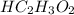 in vinegar 5.04%.
in vinegar 5.04%.