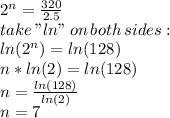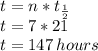Chemistry, 09.01.2020 03:31 mikaelalcool1
Me i need to turn this in really !
1. a chemical equation is shown below. hgs + o2 → hgo + so2 what are the coefficients that should be added to balance this equation? use complete sentences to explain your answer. explain how this chemical reaction demonstrates the conservation of mass.
2. part 1: name the type of chemical reaction that occurs when aluminum (al) reacts with copper nitrate (cu(no3)2).
part 2: explain why aluminum does not react with potassium nitrate (kno3) although it reacts with copper nitrate.
3.
the table shows the amount of radioactive element remaining in a sample over a period of time.
radioactive decay rate
amount of radioactive sample
(grams) time
(hours)
186.0 0
147.0 7
117.0 14
93.0 21
23.3 63
part 1: what is the half-life of the element? explain how you determined this.
part 2: how long would it take 320 g of the sample to decay to 2.5 grams? show your work or explain your answer.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 18:00
When the following equation is balanced using the smallest possible integers, what is the coefficent of oxygen gas? c7h16o(g) + o2(g) → co2(g) + h2o(g) -1 -5 -8 -16 -21
Answers: 3

Chemistry, 22.06.2019 03:00
Zoe is investigating the composition of substance a, an unknown substance. using chemical processes, she analyzes substance a and determines it is composed of sodium, oxygen, and hydrogen atoms in a ratio of 1 : 1 : 1. what is substance a? a. a compound b. an element c. a heterogeneous mixture d. a homogeneous mixture
Answers: 1

Chemistry, 22.06.2019 21:30
Plzz a sample of table sugar (sucrose, c12h22o11) has a mass of 7.801 g. ● a) calculate the number of moles of c12h22o11 in the sample b) calculate the number of moles of each element in c12h22o11 (number of moles of c, number of moles of h & number of moles of o) in the sample. (use your answer from part a as your starting point.) show your work and highlight your final answer. calculate the number of atoms of each element in c12h22o11 (number of atoms of c, number of atoms of h & number of atoms of o) in the sample. (use your answers from part b as your starting for each element.) show your work and highlight your final answer.
Answers: 1

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
Me i need to turn this in really !
1. a chemical equation is shown below. hgs + o2 → hg...
1. a chemical equation is shown below. hgs + o2 → hg...
Questions




Physics, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00

Physics, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00

English, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00

Health, 03.07.2021 14:00


Mathematics, 03.07.2021 14:00

Computers and Technology, 03.07.2021 14:00

Mathematics, 03.07.2021 14:00



Mathematics, 03.07.2021 14:00

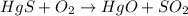
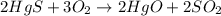
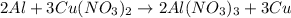
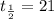 hours.
hours. --- (A)
--- (A)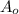 = initial mass of the sample = 320 g
= initial mass of the sample = 320 g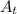 = Mass left after decaying (or after t amount of time) = 2.5 g
= Mass left after decaying (or after t amount of time) = 2.5 g