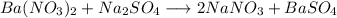Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 07:30
Calculate the ratio of h+ ions to oh– ions at a ph = 7. find the concentration of h+ ions to oh– ions listed in table b of your student guide. then divide the h+ concentration by the oh– concentration. record this calculated ratio in table a of your student guide. compare your approximated and calculated ratios of h+ ions to oh– ions at a ph = 7. are they the same? why or why not? record your comparison in table a. what is the concentration of h+ ions at a ph = 7? mol/l what is the concentration of oh– ions at a ph = 7? mol/l what is the ratio of h+ ions to oh– ions at a ph = 7? : 1
Answers: 1

Chemistry, 22.06.2019 12:00
Explain what happens at the saturation point when adding salt to water at room temperature.
Answers: 1

Chemistry, 22.06.2019 13:30
An animal cell loses the ability to convert energy stored in food to energy that the cell can use. which of the cell's organelles has stopped working? a.the mitochondria b.the nucleus c.the vacuoles d.the endoplasmic reticulum
Answers: 1

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
The reaction ba(no3)2(aq) + na2so4(aq) → 2nano3(aq) + baso4(s) goes to completion because a...
Questions

History, 12.10.2020 20:01




Mathematics, 12.10.2020 20:01

Mathematics, 12.10.2020 20:01


Mathematics, 12.10.2020 20:01

Mathematics, 12.10.2020 20:01



Mathematics, 12.10.2020 20:01


Mathematics, 12.10.2020 20:01

English, 12.10.2020 20:01

Mathematics, 12.10.2020 20:01

Mathematics, 12.10.2020 20:01


Mathematics, 12.10.2020 20:01