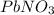Chemistry, 27.06.2019 13:00 famousshorty
The reactants of two chemical equations are listed. equation 1: pbno3 + mg equation 2: pbno3 + ki based on the type of reaction, which reaction can be used to extract lead metal from lead nitrate solution? equation 2, because k being more reactive, exchanges position with pb in pbno3 equation 2, because k being more reactive, exchanges position with n in pbno3 equation 1, because mg being more reactive, replaces n from pbno3 equation 1, because mg being more reactive, replaces pb from pbno3

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 23:00
Write a brief passage describing a neutral atom of nitrogen-14 (n-14). describe the number of protons, neutrons, and electrons in the atom, where each type of particle is located, and how the terms atomic number, mass number, and atomic mass are related to the particles. use the periodic table to you. 14 protons and eletrons since its a neutral atom
Answers: 1


Chemistry, 23.06.2019 01:30
Which conclusion fits the data in the table? a. heat chemically changes chocolate and margarine. b. all solids become liquid at 100°f. c. removing heat from a substance it to melt. d. matter may change shape when it is heated.
Answers: 1

Chemistry, 23.06.2019 09:30
What is the best describtion of the side of the moon that faces earth?
Answers: 1
You know the right answer?
The reactants of two chemical equations are listed. equation 1: pbno3 + mg equation 2: pbno3 + ki...
Questions

Mathematics, 10.07.2019 21:00


Social Studies, 10.07.2019 21:00

SAT, 10.07.2019 21:00



Chemistry, 10.07.2019 21:00


SAT, 10.07.2019 21:00

Chemistry, 10.07.2019 21:00

Advanced Placement (AP), 10.07.2019 21:00



Computers and Technology, 10.07.2019 21:00

Biology, 10.07.2019 21:00

Mathematics, 10.07.2019 21:00


Social Studies, 10.07.2019 21:10