
Gerald's science teacher mixed room temperature samples of hydrochloric acid and sodium hydroxide in a large beaker. the solution still looked clear like water, but when the students carefully touched the beaker one at a time, it felt warm to the touch. why did the beaker most likely feel warm? a. the two liquids were not soluble in water. b. the release of a gas heated the solution. c. the energy of mixing warmed the solution. d. a chemical reaction was producing a new substance.

Answers: 1
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Can you reduce your impairment by drinking coffee or cold showers? true or false?
Answers: 3

Chemistry, 22.06.2019 05:30
You are making a solution of calcium chloride dissolved in water. you add solid, stir, and it dissolves. you add just a spatula tip full, stir, and the solid does not dissolve. how could you describe the solutions before and after adding the spatula tip amount
Answers: 1

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3

Chemistry, 22.06.2019 20:20
The characteristics of two different types of reactions are shown below: reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of an element. which statement is true about the atoms of the elements that participate in the two reactions? their identity changes in both reaction a and reaction b. their identity changes in reaction a but not in reaction b. their identity changes in reaction b but not in reaction a. their identity remains the same in both reaction a and reaction b.
Answers: 1
You know the right answer?
Gerald's science teacher mixed room temperature samples of hydrochloric acid and sodium hydroxide in...
Questions

Mathematics, 11.02.2021 18:10


Mathematics, 11.02.2021 18:10


Mathematics, 11.02.2021 18:10




English, 11.02.2021 18:10


Computers and Technology, 11.02.2021 18:10

Mathematics, 11.02.2021 18:10


Mathematics, 11.02.2021 18:10

English, 11.02.2021 18:10




Mathematics, 11.02.2021 18:20

Physics, 11.02.2021 18:20


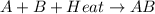
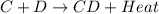 is an exothermic reaction.
is an exothermic reaction.

