
Chemistry, 26.06.2019 04:00 xxaurorabluexx
Suppose that 0.25 mole of gas c was added to the mixture without changing the total pressure of the mixture. how does the addition of c to the mixture change the mole fraction of gas a?

Answers: 2
Another question on Chemistry

Chemistry, 21.06.2019 14:00
Which of the following statements is true? question 4 options: nuclear decay rates vary with the conditions of the reaction, but chemical reaction rates do not. chemical reaction rates vary with the conditions of the reaction, but nuclear decay rates do not. neither chemical reaction rates nor nuclear decay rates vary with the conditions of the reaction. both chemical reaction rates and nuclear decay rates vary with the conditions of the reaction.
Answers: 1

Chemistry, 21.06.2019 22:10
How do forces between particles in gases compare to forces in the other states of matter? o a. the forces in gases are stronger than forces in solids but weaker than forces in liquids. o b. the forces in gases are weaker than forces in solids but stronger than forces in liquids. o c. the forces in gases are weaker than forces in solids and liquids. o d. the forces in gases are stronger than forces in solids and liquids. submit
Answers: 1


Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3
You know the right answer?
Suppose that 0.25 mole of gas c was added to the mixture without changing the total pressure of the...
Questions


Mathematics, 20.01.2021 19:30

Mathematics, 20.01.2021 19:30

Mathematics, 20.01.2021 19:30




Arts, 20.01.2021 19:30



Mathematics, 20.01.2021 19:30

Spanish, 20.01.2021 19:30


Arts, 20.01.2021 19:30



Mathematics, 20.01.2021 19:30

Mathematics, 20.01.2021 19:30


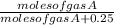 .
.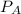 =
= 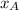 ×P (where
×P (where 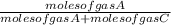 and
and 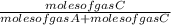 respectively.
respectively.

