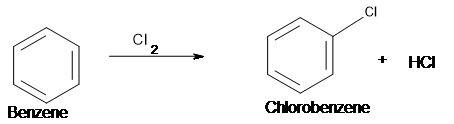
Chemistry, 25.06.2019 05:30 taliyahjhonson1
How does monochlorination work? describe with an exampel.

Answers: 1
Another question on Chemistry

Chemistry, 23.06.2019 14:00
Which of the following is not a result when a change to an equilibrium system is applied? (2 points) increasing the rate of the forward reaction will cause a shift to the left. increasing the rate of the reverse reaction will cause a shift to the left. decreasing the rate of the forward reaction will cause a shift to the left. decreasing the rate of the reverse reaction will cause a shift to the right.
Answers: 1


Chemistry, 23.06.2019 20:00
Asample of water is taken and kept in a beaker in a freezer at a constant temperature of 0°c. if the system is at dynamic equilibrium, which of these statements is true? the rate of freezing is equal to the rate of melting. the rate of freezing is greater than the rate of melting. the amount of ice is greater than the amount of water. the amount of ice is equal to the amount of water.
Answers: 1

Chemistry, 23.06.2019 22:30
The barometer shown in fig. p1.35 contains mercury ( 5 13.59 g/cm3 ). if the local atmospheric pressure is 100 kpa and g 5 9.81 m/s2 , determine the height of the mercury column, l, in mmhg and inhg.
Answers: 1
You know the right answer?
How does monochlorination work? describe with an exampel....
Questions



Mathematics, 16.12.2020 19:20

English, 16.12.2020 19:20

Social Studies, 16.12.2020 19:20


Social Studies, 16.12.2020 19:20


Biology, 16.12.2020 19:20

History, 16.12.2020 19:20

Arts, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20


Arts, 16.12.2020 19:20

Mathematics, 16.12.2020 19:20