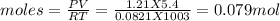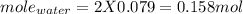Chemistry, 24.06.2019 19:30 luluhawkins
You form water vapor by mixing oxygen and hydrogen at 730°c in a 5.4-liter container. this is the equation for the reaction: o2(g) + 2h2(g) → 2h2o(g). the partial pressure of oxygen before the reaction is 122.3 kilopascals, and there is excess hydrogen. how many moles of water are formed? the reaction produces moles of water.

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

Chemistry, 22.06.2019 23:00
Which of your 24 wells had indications that a chemical reaction occurred? how were you able to tell that a chemical reaction occurred? which of your 24 wells had indications that a physical reaction occurred? how were you able to tell that a physical reaction occurred? report on both mixing and evaporation. make a general statement about whether your hypotheses were validated or rejected. must your hypotheses be correct for this to be a successful laboratory?
Answers: 3

Chemistry, 23.06.2019 04:00
Achemical reaction is done in the setup shown , resulting in a change of mass. what will happen if the same reaction is done in a sealed container that is placed on the electronic balance?
Answers: 2

Chemistry, 23.06.2019 12:40
During an experiment, ice and water were placed in a perfectly insulated thermos flask at 0 °c. describe this system when it phase reaches equilibrium.
Answers: 1
You know the right answer?
You form water vapor by mixing oxygen and hydrogen at 730°c in a 5.4-liter container. this is the eq...
Questions

History, 19.08.2019 13:00


Mathematics, 19.08.2019 13:00




Biology, 19.08.2019 13:00

Mathematics, 19.08.2019 13:00

Mathematics, 19.08.2019 13:00

Social Studies, 19.08.2019 13:00


Physics, 19.08.2019 13:00

Mathematics, 19.08.2019 13:00


Health, 19.08.2019 13:00



Mathematics, 19.08.2019 13:00


History, 19.08.2019 13:00

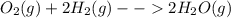
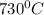 =1003K
=1003K