
Chemistry, 30.11.2019 08:31 SuperWoman9172
Carbon disulfide burns in oxygen to yield car- bon dioxide and sulfur dioxide according to the chemical equation cs2(l) 3 o2(g) −→ co2(g) 2 so2(g). if 0.91 mol of cs2 is combined with 1.52 mol of o2, identify the limiting reactant.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 12:00
Which of the following units is not an official si unit? mole liter kilogram ampere
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 19:20
The equation picture below shows which type of nuclear reaction u 235 + n x e 134 + sr 100 + 2n
Answers: 1

Chemistry, 22.06.2019 23:00
What prefix multiplier is appropriate for reporting a measurement of 5.57 ×10−5 m?
Answers: 1
You know the right answer?
Carbon disulfide burns in oxygen to yield car- bon dioxide and sulfur dioxide according to the chemi...
Questions


Mathematics, 07.01.2020 19:31



Mathematics, 07.01.2020 19:31


Computers and Technology, 07.01.2020 19:31




English, 07.01.2020 19:31

Mathematics, 07.01.2020 19:31


Mathematics, 07.01.2020 19:31


Social Studies, 07.01.2020 19:31

Mathematics, 07.01.2020 19:31

Arts, 07.01.2020 19:31


Mathematics, 07.01.2020 19:31

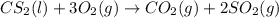
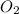 reacts with 1 mole of
reacts with 1 mole of 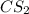
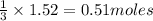 of
of 

