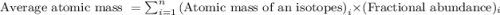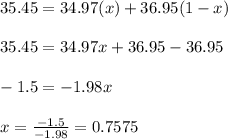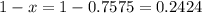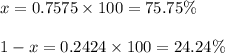Chemistry, 23.06.2019 10:30 GiuliAzevedo
The element chlorine has two stable isotopes, chlorine-35 with a mass of 34.97 amu and chlorine-37 with a mass of 36.95 amu. from the atomic weight of cl = 35.45 one can conclude that:

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 00:00
What stress will shift the following equilibrium system to the left? n2(g) + 3h2(g) ⇌ 2nh3(g) adding more n2(g) adding more nh3(g) increasing the pressure of the system reducing the volume of the container
Answers: 1

Chemistry, 22.06.2019 08:00
Which of the following observations indicates that there is a small, dense, positively charged part in the center of an atom? some uncharged particles are scattered by a gold foil. all uncharged particles are attracted towards a gold foil. all positively charged particles pass straight through a gold foil. some positively charged particles bounce back from a gold foil.
Answers: 2

Chemistry, 22.06.2019 17:30
The polymer used for the nonstick surface of cooking utensils is 24.0%c and 76%f by mass. what is the empirical formula of this polymer?
Answers: 2

Chemistry, 22.06.2019 22:00
Choose all the answers that apply. fluorine (f) has an atomic number of 9 and an atomic weight of 18.99. fluorine has a. 9 protons b. 10 neutrons c. 18 electrons d. an atomic mass of 19 e. at least one isotope
Answers: 1
You know the right answer?
The element chlorine has two stable isotopes, chlorine-35 with a mass of 34.97 amu and chlorine-37 w...
Questions

History, 06.11.2020 22:50

History, 06.11.2020 22:50


History, 06.11.2020 22:50

History, 06.11.2020 22:50



Biology, 06.11.2020 22:50

Mathematics, 06.11.2020 22:50

History, 06.11.2020 22:50

Mathematics, 06.11.2020 22:50

Mathematics, 06.11.2020 22:50


English, 06.11.2020 22:50


Mathematics, 06.11.2020 22:50



History, 06.11.2020 22:50

Arts, 06.11.2020 22:50