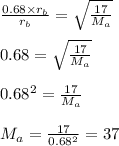Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 08:00
Why is the bond angle in a water molecule less than the bond angle of methane? a. the central oxygen atom in water has two lone pairs of electrons, whereas the central carbon atom in methane has no lone pairs. b. the central hydrogen atom in water has one lone pair of electrons, whereas the central carbon atom in methane has two lone pairs. c. the central oxygen atom in water has four lone pairs of electrons, whereas the central carbon atom in methane has only one lone pair. d. the central oxygen atom exerts more repulsive force on surrounding atoms than the central carbon atom in methane does. reset next
Answers: 2

Chemistry, 22.06.2019 15:00
Which are forms of frozen water? check all that apply. dew frost hail rain sleet
Answers: 2

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1

Chemistry, 23.06.2019 01:00
Who examines and coordinates the cleanup of polluted sites?
Answers: 2
You know the right answer?
6. gas a effuses 0.68 times as fast as gas b. if the molar mass of gas b is 17 g, what is the mass o...
Questions


English, 05.05.2021 20:30

English, 05.05.2021 20:30



Mathematics, 05.05.2021 20:30

Mathematics, 05.05.2021 20:30

Computers and Technology, 05.05.2021 20:30

Mathematics, 05.05.2021 20:30





Mathematics, 05.05.2021 20:30

Computers and Technology, 05.05.2021 20:30





History, 05.05.2021 20:30

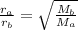
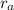 = Rate of effusion of gas A
= Rate of effusion of gas A 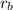 = Rate of effusion of gas B
= Rate of effusion of gas B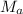 = Molar mass of gas A
= Molar mass of gas A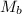 = Molar mass of gas B
= Molar mass of gas B