
Chemistry, 04.02.2020 16:58 genesisdiaz1352
Calculate the number of atoms of carbon (c) in 340. cm3 of the colorless gas methylacetylene at 0 °c and atmospheric pressure, where its density is 1.79×10-3 g cm-3. the molecular formula of methylacetylene is c3h4.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 08:30
Sally is making a model of a magnesium atom with an atomic mass number of 24 for her chemistry class. she has foam balls for the protons, neutrons, and electrons. she has added 6 neutrons to her model so far. how many more neutrons does she need to add to complete her neutral atom of magnesium?
Answers: 1

Chemistry, 22.06.2019 09:00
The diagram below shows a cell placed in a solution.a cell is shown placed inside a beaker. it is labeled cell. the solution inside the beaker is labeled 40% salt solution and the solution inside the cell is labeled 20% salt solution.only water is allowed to move in and out of the cell. what will most likely happen to the cell? it will expand as water moves out of it. it will shrink as water moves out of it.it will expand as water moves into it. it will shrink as water moves into it.
Answers: 2

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2

Chemistry, 22.06.2019 13:00
Asubstance is a good conductor of electricity which of the following best explains a probable position of the substance in a periodic table
Answers: 3
You know the right answer?
Calculate the number of atoms of carbon (c) in 340. cm3 of the colorless gas methylacetylene at 0 °c...
Questions




Mathematics, 23.10.2020 19:10

Mathematics, 23.10.2020 19:10


Social Studies, 23.10.2020 19:10



Biology, 23.10.2020 19:10


English, 23.10.2020 19:10

Biology, 23.10.2020 19:10





History, 23.10.2020 19:10


Mathematics, 23.10.2020 19:10

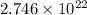
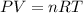
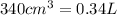 (Conversion Factor:
(Conversion Factor: 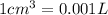 )
)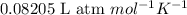
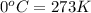 (Conversion factor:
(Conversion factor: 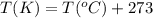 )
)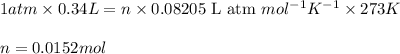
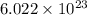 number of atoms
number of atoms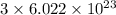 number of C-atoms.
number of C-atoms.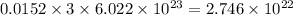 number of C-atoms.
number of C-atoms.

