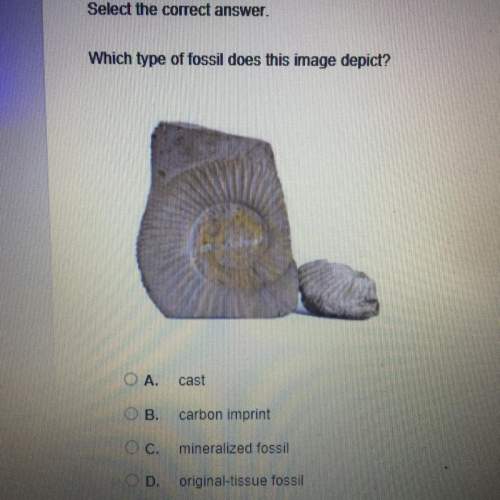
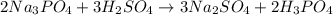Chemistry, 18.10.2019 10:30 4presidents
How do i balance this equation? sodium phosphate reacts with sulfuric acid to form sodium sulfate and phosphoric acid

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2



Chemistry, 23.06.2019 06:30
Which of the following is true about the products formed during photosynthesis? (5 points) select one: a. they have the same mass as the mass of reactants. b. they are the same set of compounds as the reactants. c. they have more mass than the mass of reactants. d. they are chemically the same as the reactants.
Answers: 1
You know the right answer?
How do i balance this equation? sodium phosphate reacts with sulfuric acid to form sodium sulfate a...
Questions









Mathematics, 12.09.2019 05:20

Business, 12.09.2019 05:20

Physics, 12.09.2019 05:20


English, 12.09.2019 05:20

Chemistry, 12.09.2019 05:20

Mathematics, 12.09.2019 05:20



Chemistry, 12.09.2019 05:20