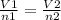Chemistry, 28.01.2020 09:31 granthazenp5e9mj
Gas laws describe and predict the behavior of gases without attempting to explain why they happen
true
false
a 6.0 l sample of nitrogen gas contains 0.50 mole of a gas. if enough gas is added to make a total of 0.75 moles at the same pressure and temperature, what is the resulting total volume of the gas? (4 points)
2.3 l
4.8 l
7.5 l
9.0 l

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 09:20
Which of these statements explains the difference between nuclear binding energy and the strong nuclear force ?
Answers: 3

Chemistry, 22.06.2019 10:30
What is the empirical formula of c6h18o3? ch3o c2h5o c2h6o c2h5o5
Answers: 1

Chemistry, 22.06.2019 14:30
Consider the reduction reactions and their equilibrium constants. cu+(aq)+e−↽−−⇀cu(s)pb2+(aq)+2e−↽−−⇀pb(s)fe3+(aq)+3e−↽−−⇀fe(=6.2×108=4.0×10−5=9.3×10−3 cu + ( aq ) + e − ↽ − − ⇀ cu ( s ) k =6.2× 10 8 pb 2 + ( aq ) +2 e − ↽ − − ⇀ pb ( s ) k =4.0× 10 − 5 fe 3 + ( aq ) +3 e − ↽ − − ⇀ fe ( s ) k =9.3× 10 − 3 arrange these ions from strongest to weakest oxidizing agent.
Answers: 3

Chemistry, 22.06.2019 21:30
How can the periodic table be used to predict the behavior of elements?
Answers: 1
You know the right answer?
Gas laws describe and predict the behavior of gases without attempting to explain why they happen
Questions

English, 02.09.2019 01:30



Biology, 02.09.2019 01:30


Mathematics, 02.09.2019 01:30





Social Studies, 02.09.2019 01:30


History, 02.09.2019 01:30


Chemistry, 02.09.2019 01:30

Social Studies, 02.09.2019 01:30


History, 02.09.2019 01:30

Mathematics, 02.09.2019 01:30

Mathematics, 02.09.2019 01:30