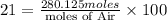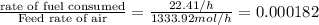Chemistry, 13.10.2019 17:20 alyxkellar06
4. gasoline, which we will represent as having the properties of isooctane, c8h18, is consumed by an idling automobile engine at a rate of 1 gal/h. co is a toxic air pollutant formed from the incomplete combustion of the gasoline. an air monitor in the garage where work is being done on an engine detects an accumulation of co. what does this information imply about the gasoline-to-air ratio being fed to the engine? if we assume that gasoline has properties of isooctane, estimate the feed rate (mol/h) of air for 10% excess oxygen fed to the engine. the mw and density of isooctane is 114.23 g/mol and 691.87 kg/m3, respectively. air contains 21 mol % o2. (hint: balance the stoichiometric equation to determine the theoretical number of moles of o2, c8h18 + n o2 = n h2o + n co2).

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 03:30
The atomic radius of sodium is 186 pm and of chlorine is 100 pm. the ionic radius for na+ is 102 pm and for cl– is 181 pm. in going from na to cl in period 3, why does the atomic radius decrease while the ionic radius increases? a. the inner electrons in the sodium cation shield its valence electrons more effectively than the inner electrons in the chloride anion do. b. the inner electrons shield the valence electrons more effectively in the chlorine atom than in the chloride anion. c. the outermost electrons in chloride experience a smaller effective nuclear charge than those in the sodium cation do. d. the outermost electrons in chloride experience a larger effective nuclear charge than those in the sodium cation do. e. monatomic ions are bigger than the atoms from which they are formed.
Answers: 2

Chemistry, 22.06.2019 12:30
Suppose you wanted to make 100 grams of water. what is the molar mass of water (h2o)?
Answers: 2

Chemistry, 22.06.2019 13:10
Which electron configuration represents the electrons in an atom of sodium in the ground state at stp
Answers: 1

You know the right answer?
4. gasoline, which we will represent as having the properties of isooctane, c8h18, is consumed by an...
Questions



Mathematics, 13.12.2021 07:10



Chemistry, 13.12.2021 07:10

Spanish, 13.12.2021 07:10

Social Studies, 13.12.2021 07:10



Advanced Placement (AP), 13.12.2021 07:10




Mathematics, 13.12.2021 07:10

English, 13.12.2021 07:10


Chemistry, 13.12.2021 07:10


Mathematics, 13.12.2021 07:10

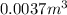
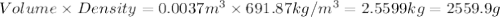
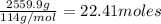
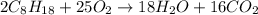
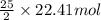 of oxygen that is 280.125 moles.
of oxygen that is 280.125 moles.