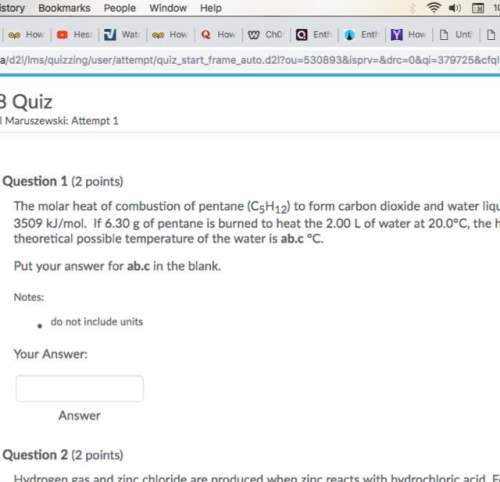
Question number 1 they are asking for highest possible theoretical temperature?
...

Chemistry, 25.01.2020 13:31 jamaicalove2880
Question number 1 they are asking for highest possible theoretical temperature?

Answers: 2
Another question on Chemistry

Chemistry, 22.06.2019 19:10
Astudent completes a titration by adding 12.0 milliliters of naoh(aq) of unknown concentration to 16.0 milliliters of 0.15 m hcl(aq). what is the molar concentration of the naoh(aq)? 1)5.0 m 2)0.20 m 3)0.11 m 4)1.1 m
Answers: 1

Chemistry, 22.06.2019 20:10
The lattice enthalpy (formation of ionic solid from ions in the gas phase) for agcl(s) is -916 kj/mol and the hydration enthalpy (dissolution of gaseous ions into water) is -850 kj/mol. how much heat (in joules) is involved in forming 1l of saturated agcl solution (1.8 × 10-4 g / 100 ml water) by dissolving agcl(s)? assume solution volume does not change much upon dissolution. the equations are given below. ag+(g) + cl−(g) æ agcl(s)
Answers: 3

Chemistry, 23.06.2019 00:30
Gasoline has a density of 0.740 g/ml. if you have 328 grams of gasoline, what is the volume in milliliters?
Answers: 1

Chemistry, 23.06.2019 02:00
Now look at the segment of the graph between the two data points marked with black squares. describe how the boiling point and melting point plots behave between these points. be as specific as possible.
Answers: 1
You know the right answer?
Questions


Health, 18.03.2021 01:40

Mathematics, 18.03.2021 01:40


History, 18.03.2021 01:40





Computers and Technology, 18.03.2021 01:40




Health, 18.03.2021 01:40

Arts, 18.03.2021 01:40


Mathematics, 18.03.2021 01:40

Mathematics, 18.03.2021 01:40


Biology, 18.03.2021 01:40



