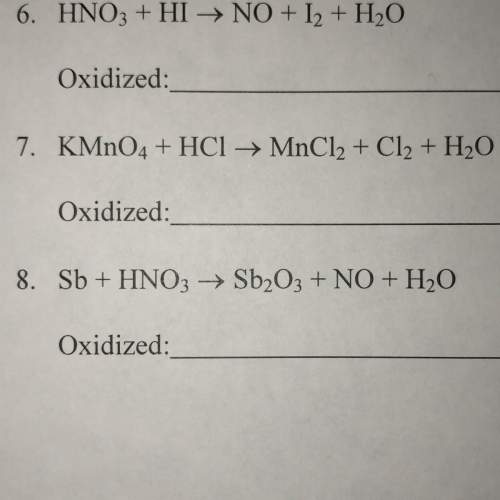
How to oxidize and reduce the following reactions
...

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 03:20
Which type of substance ionizes partially and gives off hydrogen ions when dissolved in water? a. strong acid b. strong base c. weak acid d. weak base
Answers: 1

Chemistry, 22.06.2019 22:30
3.09 lab: reaction of metals 1 which combinations of substances resulted in a chemical change? for each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. make a general statement about the reactivity of the metals in this experiment.
Answers: 1

Chemistry, 23.06.2019 00:30
Titration reveals that 11.6 ml of 3.0m sulfuric acid are required to neutralize the sodium hydroxide in 25.00ml of naoh solution. what is the molarity of the naoh solution?
Answers: 1

Chemistry, 23.06.2019 01:30
Which statement justifies that hydrogen peroxide (h2o2) is a polar molecule? the o – h bond is nonpolar and the molecule is asymmetric. the o – h bond is nonpolar and the molecule is symmetric. the o – h bond is polar and the molecule is asymmetric. the o – h bond is polar and the molecule is symmetric.
Answers: 1
You know the right answer?
Questions


Mathematics, 03.03.2021 08:20


History, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20



Mathematics, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20

Physics, 03.03.2021 08:20

Mathematics, 03.03.2021 08:20


Biology, 03.03.2021 08:20

Social Studies, 03.03.2021 08:20



Mathematics, 03.03.2021 08:20


History, 03.03.2021 08:20