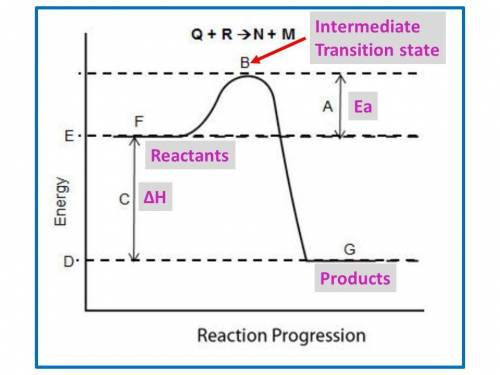
Chemistry, 08.10.2019 10:30 savid88061
The balanced equation below represents the reaction between a 5.0 gram sample of zinc metal and 0.5 m of hydrochloric acid. the reaction takes place in an open test tube at 298k and 1 atm in a laboratory activity


Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 03:40
Chemical kinetics what was the rate of reaction in trial 3? choose the closest answer.
Answers: 3

Chemistry, 22.06.2019 17:10
Some liquids can be distilled, but only at temperatures that are so high that it is impractical, or so high the compound decomposes. explain why distillation such compounds at significantly less than atmospheric pressure (some degree of vacuum) would solve this problem.
Answers: 2

Chemistry, 22.06.2019 20:00
The picture represents the process that produces most of the energy used by living organisms on earth. which process is represented in the picture? a) the magnetic attraction between two hydrogen nuclei. b) the fusion of hydrogen nuclei to produce a helium nucleus in the core of the sun. c) the fission of hydrogen nuclei to produce a helium nucleus in the core of the sun. d) the chemical reaction between hydrogen nuclei to produce a helium nucleus in earth's atmosphere.
Answers: 3

Chemistry, 23.06.2019 04:00
Calculate the mass of 0.750 mol of the following substance. na3po4. , i'm not quite sure on how to set up the problem to solve! : (
Answers: 1
You know the right answer?
The balanced equation below represents the reaction between a 5.0 gram sample of zinc metal and 0.5...
Questions


Physics, 09.01.2020 15:31





Mathematics, 09.01.2020 15:31

Biology, 09.01.2020 15:31


Mathematics, 09.01.2020 15:31




Mathematics, 09.01.2020 15:31


Biology, 09.01.2020 15:31




Biology, 09.01.2020 15:31