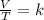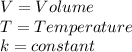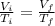Chemistry, 09.11.2019 11:31 cillalynn24
Which law states that the volume and absolute temperature of a fixed quantity of gas are directly proportional under constant pressure conditions?
boyle’s law
charles’s law
dalton’s law
gay-lussac’s law

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:00
Which part of a feedback mechanism is able to monitor the conditions outside of cells and usually uses nerve cells to relay this information to an intergrating center
Answers: 2

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3


Chemistry, 22.06.2019 17:40
If 3 moles of a compound use 24 j of energy in a reaction, what is the a hreaction in j/mol?
Answers: 1
You know the right answer?
Which law states that the volume and absolute temperature of a fixed quantity of gas are directly pr...
Questions





Mathematics, 29.01.2021 18:30

Chemistry, 29.01.2021 18:30

English, 29.01.2021 18:30

Biology, 29.01.2021 18:30


Mathematics, 29.01.2021 18:30

Biology, 29.01.2021 18:30

Mathematics, 29.01.2021 18:30


Mathematics, 29.01.2021 18:30

Mathematics, 29.01.2021 18:30

Health, 29.01.2021 18:30

Social Studies, 29.01.2021 18:30


Mathematics, 29.01.2021 18:30