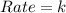Chemistry, 05.02.2020 02:50 damienlopezram
What is the overall order of reaction for this rate law: rate = k ? a. zero b. first c. second d. third

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 01:30
An empty fuel tank can still contain and therefore can be even more dangerous than one full of liquid fuel.
Answers: 1

Chemistry, 22.06.2019 07:30
The volume of helium in a blimp is 6.28 x 10^9 millimeters. the density of helium in the blimp is .1786 kilogram/meter^3. find the mass of the helium in the blimp.
Answers: 1

Chemistry, 22.06.2019 09:40
Consider this initial-rate data at a certain temperature for the reaction described by
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
What is the overall order of reaction for this rate law: rate = k ? a. zero b. first c. second d....
Questions


English, 15.09.2021 14:00

Mathematics, 15.09.2021 14:00


Mathematics, 15.09.2021 14:00


Computers and Technology, 15.09.2021 14:00


Mathematics, 15.09.2021 14:00

Mathematics, 15.09.2021 14:00

Social Studies, 15.09.2021 14:00

History, 15.09.2021 14:00

Mathematics, 15.09.2021 14:00


Chemistry, 15.09.2021 14:00

History, 15.09.2021 14:00

English, 15.09.2021 14:00

English, 15.09.2021 14:00

English, 15.09.2021 14:00

Mathematics, 15.09.2021 14:00