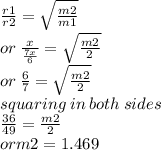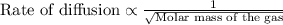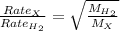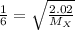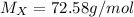Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 10:30
Consider the following reactions. (note: (s) = solid, (l) = liquid, and (g) = gas.) mg(s) + ½o2(g) → mgo(s) + 146 kcal/mole h2(g) + ½o2(g) → h2o(g), δh = -57.82 kcal/mole what type of reaction is represented by the previous two examples?
Answers: 3


Chemistry, 23.06.2019 00:00
In an exothermic reaction, energy may be released to the surroundings in the form of question 4 options: heat light thermal all of the above
Answers: 3

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1
You know the right answer?
Agas diffuses 1/6 times faster than hydrogen gas (h2). what is the molar mass of the gas? 59.95 g/m...
Questions





Mathematics, 12.02.2021 04:10

Chemistry, 12.02.2021 04:10


Chemistry, 12.02.2021 04:10

Social Studies, 12.02.2021 04:10

Mathematics, 12.02.2021 04:10


Mathematics, 12.02.2021 04:10

Chemistry, 12.02.2021 04:10

Mathematics, 12.02.2021 04:10

Mathematics, 12.02.2021 04:10


Computers and Technology, 12.02.2021 04:10

Computers and Technology, 12.02.2021 04:10