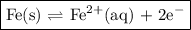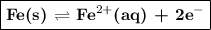Chemistry, 30.06.2019 02:40 ComicSans10
The half-cell is a chamber in the voltaic cell where one half-cell is the site of the oxidation reaction and the other half-cell is the site of the reduction reaction. type the half-cell reaction that takes place at the anode for the iron-silver voltaic cell. indicate the physical states of atoms and ions using the abbreviation (s), (l), or (g) for solid, liquid, or gas, respectively. use (aq) for an aqueous solution. do not include phases for electrons. express your answer as a chemical equation.

Answers: 3
Another question on Chemistry

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 14:30
Chemistry worksheet - i am not sure what they are asking for exactly?
Answers: 1

Chemistry, 22.06.2019 16:40
Identify the lewis acid in this balanced equation: ag+ + 2nh3 -> ag(nh3)2+a. ag+b. nh3c. ag(nh3)2+
Answers: 1

Chemistry, 22.06.2019 17:30
Air can be considered a mixture. which statement does not explain why?
Answers: 1
You know the right answer?
The half-cell is a chamber in the voltaic cell where one half-cell is the site of the oxidation reac...
Questions




History, 23.09.2019 12:30

Mathematics, 23.09.2019 12:30

Social Studies, 23.09.2019 12:30

History, 23.09.2019 12:30

Social Studies, 23.09.2019 12:30

Mathematics, 23.09.2019 12:30


Biology, 23.09.2019 12:30


Biology, 23.09.2019 12:30


History, 23.09.2019 12:30

Computers and Technology, 23.09.2019 12:30


Mathematics, 23.09.2019 12:30

Mathematics, 23.09.2019 12:30

English, 23.09.2019 12:30