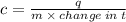When 45 g of an alloy, at 25°c, are dropped into 100.0g of water, the alloy absorbs 956j of heat. if the temperature of the alloy is 37°c, what is its specific heat? a. 0.423 cal/g°c b. 1.77 cal/g°c c. 9.88 cal/g°c d. 48.8 cal/g°c try and explain with step by step or show work,

Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 00:40
During which time interval does the object travel approximately 10 meters
Answers: 3

Chemistry, 22.06.2019 04:00
Drag each label to the correct location on the chart. classify each reaction as endothermic or exothermic.
Answers: 1

Chemistry, 22.06.2019 06:30
Summarize possible ways in which phases of matter could combine to form a solution.
Answers: 2

You know the right answer?
When 45 g of an alloy, at 25°c, are dropped into 100.0g of water, the alloy absorbs 956j of heat. if...
Questions

Mathematics, 13.07.2019 09:30

Social Studies, 13.07.2019 09:30

Social Studies, 13.07.2019 09:30

Mathematics, 13.07.2019 09:30

Mathematics, 13.07.2019 09:30


Biology, 13.07.2019 09:30

Social Studies, 13.07.2019 09:30

History, 13.07.2019 09:30

Mathematics, 13.07.2019 09:30

Mathematics, 13.07.2019 09:30


Social Studies, 13.07.2019 09:30

Biology, 13.07.2019 09:30

Biology, 13.07.2019 09:30

Mathematics, 13.07.2019 09:30




Social Studies, 13.07.2019 09:30