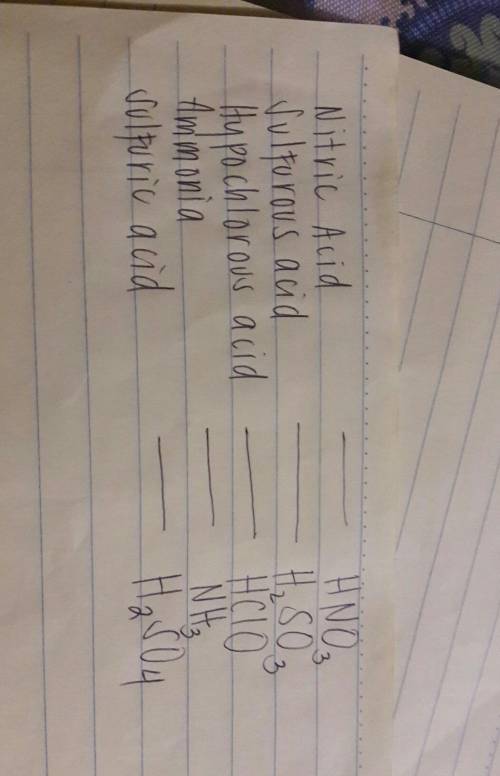Answers: 1
Another question on Chemistry

Chemistry, 22.06.2019 14:30
For the reaction shown, find the limiting reactant for each of the following initial amounts of reactants. 4al(s)+3o2(g)→2al2o3(s) a) 1 molal, 1 mol o2 b) 4 molal, 2.6 mol o2 c) 16 molal, 13 mol o2 d) 7.4 molal, 6.5 mol o2
Answers: 3

Chemistry, 22.06.2019 19:10
Δu of , in kj/kg, as it isto k, (a)as a of , (b) at , (c) at .
Answers: 2

Chemistry, 22.06.2019 20:00
Iam hoping to create 5.72 grams of glucose. the plant was given 4.75 liters of co2 and 2.81 g of h20. which reactant was the limiting reagent? how much excess mass did we have of the other reactant?
Answers: 1

Chemistry, 22.06.2019 20:30
How many grams of phosphorus are contained in 5.09 moles of phosphorus?
Answers: 1
You know the right answer?
Drag each tile to the correct box. match the name of each compound to its chemical formula. use the...
Questions

Biology, 20.08.2019 10:50


Mathematics, 20.08.2019 10:50




History, 20.08.2019 10:50

Mathematics, 20.08.2019 10:50




Chemistry, 20.08.2019 11:00

Biology, 20.08.2019 11:00


Chemistry, 20.08.2019 11:00

Mathematics, 20.08.2019 11:00


Mathematics, 20.08.2019 11:00